Baby Has Rsv Could Mother Using Vapor Cigarette
- Research article
- Open up Admission
- Published:
Systematic literature review assessing tobacco fume exposure every bit a hazard factor for serious respiratory syncytial virus disease among infants and young children
BMC Pediatrics book 12, Commodity number:81 (2012) Cite this article
Abstruse
Background
The function of environmental tobacco smoke (ETS) exposure equally a risk gene for serious respiratory syncytial virus (RSV) affliction among infants and young children has not been clearly established. This systematic review was conducted to explore the association between ETS exposure and serious RSV disease in children younger than 5 years, including infants and young children with elevated risk for serious RSV affliction.
Methods
A systematic review of English-language studies using the PubMed and EMBASE databases (1990–2009) was performed to retrieve studies that evaluated ETS as a potential gamble factor for serious RSV illness. Studies assessing gamble factors associated with hospitalization, emergency department visit, or md visit due to RSV (based on laboratory confirmation of RSV or clinical diagnosis of RSV) in children nether the age of 5 years were included.
Results
The literature search identified thirty relevant manufactures, categorized by laboratory confirmation of RSV infection (n = 14), clinical diagnosis of RSV disease (n = 8), and cess of RSV affliction severity (n = eight). Across these iii categories of studies, at least ane type of ETS exposure was associated with statistically meaning increases in risk in multivariate or bivariate analysis, as follows: 12 of fourteen studies on hazard of hospitalization or ED visit for laboratory-confirmed RSV infection; 6 of 8 studies of RSV affliction based on clinical diagnosis; and 5 of the eight studies assessing severity of RSV equally shown past hospitalization rates or degree of hypoxia. Also, 7 of the 30 studies focused on populations of premature infants, and the majority (5 studies) found a pregnant association betwixt ETS exposure and RSV risk in the multivariate or bivariate analyses.
Decision
We found ample bear witness that ETS exposure places infants and young children at increased risk of hospitalization for RSV-attributable lower respiratory tract infection and increases the severity of illness among hospitalized children. Boosted evidence is needed regarding the association of ETS exposure and outpatient RSV lower respiratory tract disease. Challenges and potential pitfalls of assessing ETS exposure in children are discussed.
Groundwork
Nearly all children contract respiratory syncytial virus (RSV) by 2 years of historic period [1]. RSV causes upper airway infections, bronchiolitis, wheezy bronchitis, and pneumonia. Although near infections produce balmy disease, RSV is a major cause of hospitalization in infants [ii] and can exist fatal [3]. Premature infants, infants with bronchopulmonary dysplasia (BPD) (chronic lung disease of prematurity), and infants with congenital heart disease are at risk for serious infections [4, v]. Risk factors for RSV infection include sectional canteen feeding [6, 7], having older brothers or sisters in the household [half dozen, 8, 9], male gender [10], low nativity weight [10, 11], prematurity [half dozen], household crowding [12], and young chronologic age [9]. Although exposure to environmental tobacco fume (ETS) is a risk factor for asthma, wheezing, decreased pulmonary function, otitis media, cough, and lower respiratory tract infections (LRTIs) in full general [13], its role in the development of serious RSV illness among infants and young children is less articulate and has been a topic of interest among the healthcare community [14].
In this systematic review, we evaluated the evidence of an association betwixt ETS exposure and serious RSV illness among children younger than 5 years. To obtain the broadest understanding of whether ETS exposure affects the adventure of serious RSV disease, we placed no limits on the type of proxy measures of ETS exposure. Understanding the evidence basis for ETS exposure and RSV could highlight the need to directly healthcare resources or intervention programs toward this potentially modifiable risk factor.
Methods
Patient population
In this systematic literature review, we included studies of infants and children up to 5 years of age, including studies of children at high risk for serious RSV disease. We defined the high-risk population as patients with prematurity, BPD, or congenital centre disease. All other patient populations were defined as general populations, which consisted predominantly of children not defined as loftier run a risk.
Search strategy
Following PRISMA guidelines, we conducted a systematic review of the association of ETS exposure with serious RSV illness by searching broadly for studies assessing various risk factors, including ETS exposure, for RSV, bronchiolitis, or LRTI caused by RSV or bronchiolitis.[15] We searched the PubMed (including MEDLINE) and EMBASE databases for English-language studies published and indexed between 1990 and April 2009. No additional relevant unpublished studies were obtained. Studies that identified ETS exposure as a hazard factor might exist more likely to mention ETS in the title or abstract than studies that evaluated ETS exposure in a set of other risk factors, only did not find information technology to exist significantly associated with increased risk. To minimize article selection bias beyond studies, we searched for articles that assessed any adventure factors for RSV also equally bronchiolitis, considering 50% to 80% of winter bronchiolitis is due to RSV in infants and immature children [1]. For PubMed, the National Library of Medicine Medical Subject Headings (MeSH terms) used in the search included tobacco smoke pollution/adverse furnishings respiratory syncytial virus infections bronchiolitis viral, and respiratory tract infections in combination with the MeSH subheadings epidemiology etiology, and complications. The MeSH heading respiratory tract infections was combined with the keywords syncytial or RSV OR bronchiolitis or the MESH term tobacco smoke pollution/adverse effects. Studies that included hazard cess were identified using the following MeSH headings: assay of variance (which includes multivariate assay), probability (which includes proportional hazards model, odds ratio, take a chance, run a risk assessment, and hazard factors); and case–control studies or accomplice studies, which were combined with the terms relative hazard OR hazard ratio OR odds ratio. The search identified studies with the following combination of these terms: (1) any disease-related term plus any risk assessment term; or (two) whatsoever disease-related term plus the fume exposure term. Nosotros did non search for unpublished studies or reports.
Report selection
Each relevant article was read by several authors and the bibliographies of included manufactures were reviewed for additional studies. We excluded studies that did non assess disease risk or did not include ETS exposure as a adventure factor. Figure one presents a summary of exclusions and rationales for exclusion during successive rounds of review.
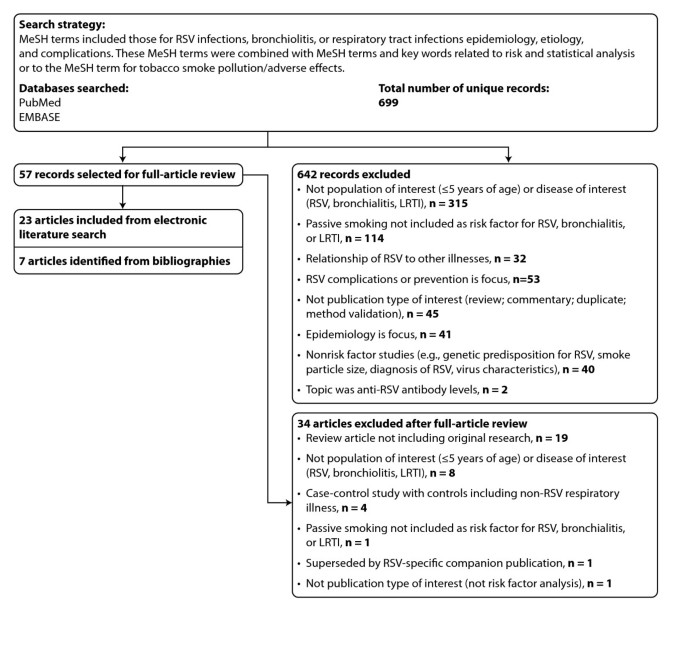
PRISMA Flow Diagram: Summary of Exclusions and Rationales for Exclusion During Phases of Systematic Literature Review. PRISMA = Preferred Reporting Items for Systematic Reviews and Meta-Analyses; LRTI = lower respiratory tract infection; MeSH = medical subject headings; RSV = respiratory syncytial virus.
Data extraction
Study details were extracted into tables, and table content was verified by a 2d writer and by a reviewer not involved in the information extraction. The authors discussed each article to achieve consensus regarding the report details. For each study the post-obit data were extracted: reference, publication twelvemonth, country origin, study pattern, study population size and description, assessment method for ETS exposure (due east.g., maternal smoking through nascency document information), method of ascertainment of RSV status (if any, such as through laboratory confirmation or clinical diagnosis), disease effect evaluated (eastward.1000., hospitalization for RSV), and results and significance of multivariate, bivariate, and other statistical analyses. Outcomes related to the presence of RSV antibodies without show of hospitalization, ED visit, or doc visit related to RSV were non extracted. The principal summary measures were adjusted odds ratios (aORs), adjusted rate ratios (aRRs), or adjusted hazard ratios (aHRs) for multivariate analyses and odds ratios (ORs) for bivariate analyses. Several studies that did not perform multivariate analyses performed betwixt-group comparisons and presented P values.
Assessment of adventure of bias
Among the studies meeting the inclusion criteria, nosotros evaluated the risk of bias at the outcome and study level using the Cochrane risk of bias tool [16]. The Cochrane risk of bias tool was developed primarily for use in interventional studies; we used the tool to examine factors specific to sources of bias frequently institute in observational studies. The studies were examined for risk of bias that would potentially influence the clan of ETS exposure with serious RSV illness. The studies were judged regarding evidence of misclassification bias of RSV disease, selective reporting bias, confounding bias, exposure ascertainment bias, or participant pick bias. Disease misclassification bias was assessed based on the likelihood of the study population having an LRTI that was not due to RSV. We await that disease misclassification would underestimate the clan betwixt ETS exposure and serious RSV illness. Selective reporting bias was evaluated based on whether outcomes related to hazard factors were clearly reported. Confounding bias was assessed based on whether the study controlled for the effects of other variables through multivariate analysis. We expect that unadjusted results may overestimate the clan between ETS exposure and serious RSV disease. Exposure observation bias was assessed based on whether the ETS exposure variables were described clearly and determined past the written report authors to be adequate measures of ETS exposure in the study population. Finally, participant pick bias was pointed out in case–control studies in which the control group consisted of ill patients with non-RSV respiratory illness, and therefore, the effect of ETS exposure on serious RSV illness could be biased toward the null.
Results
The search of PubMed and embase databases yielded 699 unique results, and the abstracts were reviewed for relevance (Figure i). Of these, 676 were excluded through successive rounds of review, the majority because the studies did not focus on a disease of interest (RSV, bronchiolitis, or LRTI attributable to RSV or bronchiolitis), were not conducted in a population of interest (children younger than 5 years without serious nonrespiratory illness, such as cancer or organ transplant), or did non assess ETS exposure.
Full review of 57 articles resulted in the exclusion of 34 articles, leaving 23 manufactures identified by the electronic search. The addition of 7 articles identified from references cited (4 were published before 1990) produced a total of thirty relevant articles.
Overview of studies
The results are organized past study characteristics and include a bias assessment summary for each study. (The full bias assessment is presented in Additional file 1: Table S1) Table 1 presents 14 studies that established a diagnosis of RSV disease by confirmatory laboratory testing. These studies compared children with RSV infections with children without respiratory affliction and assessed whether ETS exposure increased the run a risk for developing a serious RSV infection. Table two presents viii studies that did non confirm suspected cases of RSV disease with laboratory testing, but relied on the clinical diagnosis of RSV or bronchiolitis, frequently from medical charts or insurance data. These studies also compared children with RSV or bronchiolitis infections with children without respiratory disease. Tabular array iii presents 8 studies that examined the impact of ETS exposure on the severity of RSV illness as measured by hospitalization or caste of hypoxia. These studies started with a sample of children infected with RSV and assessed whether ETS exposure increased the severity of the RSV infection. Within each group of studies, we considered studies of premature infants separately considering this population is at higher run a risk for serious RSV disease than the general population of children [17, 18]. We also present the results by study design (e.g., cohort, instance–control) in each tabular array.
The methods used to assess ETS exposure condition varied widely and included mother's prenatal smoking status, female parent's postnatal smoking status, father's smoking status, smoking in the habitation, number of smokers in the household, number of cigarettes smoked in the home, more than five cigarettes smoked per day in the dwelling house, days of fume exposure, history of exposure to smoking, smoking past daycare provider, and cotinine levels.
Some of the studies summarized included more than one source of ETS exposure, but no study detailed missing ETS exposure condition data. In the studies reporting multiple ETS exposure, we have presented all results based on the dissimilar sources (e.g., maternal smoking during pregnancy and smokers in the household) because it is hard to determine in each setting which of several proxies for total ETS exposure results in the to the lowest degree misclassification error. Few of the multivariate studies provided a rationale for their selection of control variables. We therefore included positive bivariate results in our tables when bivariate and multivariate analyses produced disparate conclusions.
ETS exposure and laboratory-confirmed RSV disease
The studies in Tabular array 1 seek to reply the question, does ETS exposure increase the risk that an uninfected child will develop serious RSV? In fourteen studies, the diagnosis of RSV infection was confirmed past laboratory testing (Table 1). Of these, 12 studies showed a significant agin impact of ETS exposure on serious RSV in bivariate or multivariate analysis as measured past at least 1 exposure variable. These studies examined the association of ETS exposure and other factors on the risk of ED visit [23] or hospital admission [5, 6, 9, 10, 12, 19–21, 24–26] for laboratory-confirmed RSV LRTI. Two studies examining laboratory-confirmed RSV did not find a statistically significant association between ETS exposure and serious RSV: a cohort study of outpatients in the United States assessing the risk of a child presenting to the pediatrician'southward function with an RSV infection [22], and an Australian case–control study assessing the take chances of RSV hospitalization [11].
Four of these xiv studies contained at least 1 pregnant association, simply found mixed results with different ETS exposure variables [vi, 19, xx, 23]. One prospective cohort study in premature infants establish that maternal smoking and parental smoking in the domicile had conflicting associations with iii different outcomes: RSV LRTI, all-cause hospital admission, and length of hospital stay [nineteen]. Measures of smoke exposure in the home and maternal smoking gave alien results when evaluated in 2 studies [half-dozen, xx], including a report in premature infants [xx]. Finally, a pocket-size case–command study in a general population in Turkey establish that several exposure variables (simply mother smoking, both parents smoking, whatever parent smoking, and serum cotinine) were associated with significant increase in risk of admittance to the ED; however, only father smoking was significantly associated with a subtract in risk [23].
Of the studies of laboratory-confirmed RSV hospitalization described in Table 1, v were conducted in premature infants and 9 were conducted in general populations. All 5 studies of laboratory-confirmed RSV in premature infants found increased run a risk related to ETS exposure in either bivariate or multivariate analyses [9, 10, 19–21] (Table one). The 2004 example–control written report of infants with a gestational age of 33 to 35 weeks by Figueras-Aloy et al. [21] constitute an OR of one.62 in the bivariate analysis that did not remain significant in the multivariate analysis. However, the same researchers published a much larger accomplice study in 2008, in a premature population with a gestational age of 32 to 35 weeks, reporting a nearly identical aOR (1.61) for maternal smoking that remained significant in the multivariate analysis [20]. The authors of the 2004 written report notation that the prevalence of ETS exposure decreased in Spain during the years before the study (52% during the 1999–2000 RSV flavor compared with 30% in this study) considering of anti-smoking campaigns or wellness education [21]. The pregnant association in the larger written report suggests that the earlier study was underpowered for the multivariate assay.
Of the 9 studies of laboratory-confirmed RSV illness in full general populations described in Tables 1 2 were prospective accomplice studies and 7 were instance–command studies (Table 1). Risk of RSV LRTI in an outpatient role setting [22] and risk of RSV hospitalization from ETS exposure (smoking in the household, but not from maternal smoking during pregnancy) [6] were not significant in the cohort studies.
The 7 case–control studies of laboratory confirmed RSV affliction in general populations ranged in size from 20 to 2,564 cases (Table 1), and all found a significant association between ETS exposure and risk of RSV hospitalization in either the multivariate or bivariate analyses, with the ORs from the 4 largest studies clustering tightly between 1.35 and 1.half dozen. The 3 smallest case–control studies included in Table 1 did not provide ORs but compared ETS exposure prevalence between the cases and healthy controls [23–25]. All 3 found significant differences in ETS exposure in cases versus controls. The Turkish study also demonstrated significantly higher cotinine levels in serum samples from cases than from good for you controls [23]. In a example–control study of Alaska native children, take a chance was apparent in the bivariate assay, only not in the multivariate analyses [12] (Tabular array 1). The authors noted that misclassification of exposure status may accept been a problem. The study was conducted in Alaska, where children spend much fourth dimension indoors during the winter RSV season and could be exposed to ETS when visiting other houses or community buildings. This appears probable, considering there were smokers living in the households of 59% of the controls and 68% of the cases. The consistency of findings associating ETS exposure and increased risk of serious RSV, coupled with the inconsistency of the variables associated with these significant results, highlights the challenges of assessing and categorizing ETS exposure.
ETS exposure and clinically diagnosed RSV disease
As with the studies in Table 1, the studies in Table two seek to answer the question, does ETS exposure increase the risk that an uninfected kid volition develop serious RSV? In these studies, however, the RSV diagnosis was based on clinical findings rather than a laboratory test. Tabular array 2 presents 8 studies, half-dozen of which reported a significant association between ETS exposure and bronchiolitis hospitalization or outpatient treatment. Because other organisms may cause bronchiolitis, there may exist possible misclassification of RSV disease status [33].
The large general population accomplice studies including bronchiolitis hospitalization as an event past Boyce et al. [4], Carroll et al. [28], and Koehoorn et al. [29] are in understanding with the 12 studies in Table one that identified an increased risk of RSV hospitalization associated with ETS exposure. Carroll and colleagues also demonstrated increased risk of ED visit or clinic visit for bronchiolitis and bronchiolitis diagnosis related to ETS exposure [28].
In other studies assessing risk of bronchiolitis LRTI in children presenting in the role setting, two studies plant an increased take chances for at to the lowest degree one ETS variable [28, 31], whereas 2 other studies did not [29, 30]. Of these, the positive written report by Carroll et al. [28] had the greatest statistical power.
Only the Holman study assessed take chances of decease from bronchiolitis; the risk was increased by maternal smoking (aOR, 1.6) [three].
ETS exposure and severity of RSV affliction
The 8 studies in Table iii address the question, given that a child has contracted RSV, does ETS exposure increase the severity of illness? Four studies compared ETS exposure in children hospitalized for RSV and children with RSV who were non hospitalized. 3 of these studies, including a minor report of premature infants with BPD, found no effect of ETS exposure on the likelihood of hospitalization for RSV [two, 32, 34]. One small study found an effect of ETS exposure, just only 40% of the hospitalized children were constitute to take RSV infections, with the remaining lx% representing bronchiolitis caused by other viruses [33].
Four studies examined clinical severity (caste of hypoxemia) of RSV LRTIs among children hospitalized for treatment. All 4 plant that ETS exposure was associated with more severe illness (Table 3). Bradley et al. found that hypoxemia was associated with postnatal simply non prenatal smoke exposure [36]. Chatzimichael et al. found that breastfeeding was protective against the outcome of ETS exposure on disease severity [37]. Based on a small number of studies of RSV severity, ETS exposure does not appear to increase the risk of hospitalization versus outpatient treatment among children infected with RSV, but hospitalized RSV patients exposed to ETS take more severe affliction.
Summary of potential bias in observational studies of ETS exposure and serious RSV illness
As the studies reviewed were observational, we evaluated the chief sources of bias that could potentially affect the estimates of clan with ETS exposure (Additional file 2). We found that 21 of the 30 studies confirmed the RSV diagnosis through laboratory testing for RSV or multiple respiratory illnesses including RSV in all of the study populations [two, five, six, 9–12, 19–26, 32–36, 38]. The remaining studies had a risk of disease misclassification bias of RSV because diagnostic codes (primarily for bronchiolitis) or diagnoses from medical records were used [3, iv, 27–31, 37, 39]. Misclassification of RSV disease may attenuate the association of ETS exposure and severe RSV disease, and the studies of clinically diagnosed RSV patients may provide conservative estimates of ETS exposure hazard.
Overall, we identified few studies that had potential selective reporting bias, misreckoning bias, exposure observation bias for ETS, or participant selection bias. Selective result reporting was a potential problem in 2 studies [38, 39], meaning that outcomes were not described sufficiently to decide which factors were included and potentially controlled for in the assay. Although confounding is a threat beyond observational studies (because it is difficult to control for both measureable and unmeasurable factors), multivariate analyses were not conducted in a number of studies [23–25, 32, 34, 38], which could lead to an overestimate of the effect of ETS exposure. 1 written report had a high chance of participant selection bias, where both cases and controls were diagnosed with bronchiolitis [33]. Just two studies had an unclear risk of ETS exposure [12, 35]. Al-Sonboli and colleagues [35] did not describe how the ETS exposure information or other demographic data were obtained. In the Bulkow et al study, ETS exposure was highly prevalent in the community (in both cases and controls) and indoor visiting was frequent during the winter RSV flavor [12]; therefore, the ETS exposure variable "smoker in household" was non an adequate proxy for exposure in this population.
No statistical assay of the gamble of bias beyond studies was performed for this review.
Give-and-take
The impact of ETS exposure on RSV disease in infants and young children is consequent among studies using laboratory confirmation of RSV infection and clinical diagnosis of bronchiolitis or RSV. Among 14 population-based studies that examined the risk of admission to the hospital or ED for RSV disease, 12 showed that at least one type of ETS exposure in each report was associated with a significant adverse result in the bivariate or multivariate analysis. The ascertainment that ETS exposure increases the risk that a child volition develop RSV illness that will require hospitalization is robust considering these studies used different methods (prospective, retrospective, cohort, instance–command) in different patient populations (infants who were premature, term, or with compromising conditions), in a diversity of countries and cultures. In add-on, the bear witness suggests that ETS exposure is associated with more than severe hypoxia amidst children hospitalized for RSV [35–38], and one written report found an increased risk of mortality from bronchiolitis [3].
Amid studies in premature infants, 5 [9, 10, nineteen–21] of vii [ix, ten, xix–21, 27, 32] plant ETS exposure to be a significant hazard factor in bivariate or multivariate analysis, including 5 of the half dozen [ix, 10, nineteen–21, 32] studies assessing laboratory-confirmed RSV. One study contradicting this conclusion did not have laboratory confirmation of the diagnosis and relied on a claims database, rather than direct information drove, for ascertainment of the ETS exposure status [27]. Misclassification of disease condition or missed diagnosis may have contributed to the negative findings in this report. The other was a minor study of 30 premature infants with BPD on abode oxygen therapy, xvi of whom adult RSV [32].
The show concerning whether ETS exposure increases the risk of mild RSV infection is much less convincing. In a very big report, Carroll et al. found ETS exposure to be associated with only a small increased risk of RSV illness (OR, ane.06) presenting in the outpatient setting [28]. If this OR represents the true risk, the Holberg [22] and Marbury [thirty] studies, which did not find a take a chance, would have been underpowered to observe it because of smaller sample sizes. Because well-nigh all children contract RSV during the first few years of life [1] and most cases are balmy [two], in that location may exist a ceiling effect. If children with no ETS exposure are nearly sure to contract RSV, ETS exposure tin increase the risk of balmy infection only slightly.
Misclassification of ETS exposure is a major challenge in studying associations of ETS exposure with disease. Misclassification of exposure status produces a bias in the direction of reducing the apparent magnitude of the risk, leading to either an underestimate of the true ETS exposure risk or to a null finding. Misclassification of an infant or child'southward ETS exposure comes from researchers' use of 1 or a few measures of possible exposure, which actually can come through many avenues. Examples include in utero exposure through active and passive maternal smoking and postnatal smoking by the mother, father, other individuals living in the dwelling house, visitors, and babysitters. ETS exposure outside the home occurs in public places, day care settings, and houses of friends and relatives. No study in this review gathered exposure information for all potential sources. The smoking status of, for example, the parent is a poor proxy for this global exposure as demonstrated by cotinine level studies. Urinary or serum cotinine (a metabolite of nicotine) is an objective measure of ETS exposure but provides information regarding only the previous 48 to 72 hours of exposure [40]. In 1 study, infants whose parents reported that they did not smoke had mean cotinine levels that were 80% as high every bit those for infants with one smoking parent [39]. Some infants of nonsmoking parents had higher cotinine levels than some infants with two smoking parents [39].
Confounding bias is another potential obstacle to determining the association between ETS exposure and serious RSV disease. A few studies plant an ETS exposure effect in bivariate analyses just not in multivariate analyses after adjustment for other factors [11, 21, 39]. Multivariate models that include ETS exposure and its related factors may be difficult to interpret due to potential collinearity. Smoking status has a well-known association with socioeconomic status (SES) [41], and SES is predictive of ETS exposure in children [42]. Therefore, SES may be a proxy for global ETS exposure over the early years of life. In addition to SES, smoking status is as well predicted by race, educational attainment, and marital status [43]. Few of the multivariate studies provided a rationale for their choice of control variables. In reporting of future studies, greater details near the multivariate modeling steps may assist in cess of collinearity when meaning bivariate outcomes get nonsignificant in multivariate analysis.
This review has several limitations. The search was limited to studies published from 1990 to April 2009 in the English language. We searched only Pubmed and Embase and did not attempt to locate unpublished studies. The nature of the primary studies precluded a meta-analysis. The large retrospective database analyses included in this study (Table two) all depended on bronchiolitis or RSV disease classification from diagnostic codes or medical tape diagnosis. Although RSV is a leading cause of LRTI in infants and children, identifying the etiology of LRTI is non systematically undertaken in EDs or doc offices. Methods and reporting of ETS exposure ascertainment in these studies also varies widely. Yet, in most studies, data on the child's ETS exposure level ultimately are gathered from parent or caregiver report, whether through direct data collection for the study or, for example, through retrospective review of the mother'south prenatal health records. Our review highlights the inherent difficulty of accurately assessing global ETS exposure.
Determination
Overall we institute ample show that ETS exposure places infants and young children at increased risk of hospitalization for RSV-attributable LRTIs, and increases the severity of illness every bit measured by degree of hypoxia among children hospitalized for RSV. Based on a minor number of studies, we also plant evidence that ETS exposure does non increment the likelihood amongst general populations with RSV of hospitalization versus outpatient treatment.
The prevention of serious RSV illness provides i more rationale for protecting infants and young children from exposure to tobacco smoke, peculiarly loftier-take a chance groups such every bit premature infants and those with chronic conditions who are considered at increased risk of serious RSV disease.
References
-
Hall CB: Textbook of Pediatric Infectious Diseases. 1998, WB Saunders, Philadelphia, PA, 2084-2110. iv
-
Hall CB, Weinberg GA, Iwane MK, Blumkin AK, Edwards KM, Staat MA, Auinger P, Griffin MR, Poehling KA, Erdman D, et al: The burden of respiratory syncytial virus infection in young children. N Engl J Med. 2009, 360 (6): 588-598. 10.1056/NEJMoa0804877.
-
Holman RC, Shay DK, Curns AT, Lingappa JR, Anderson LJ: Gamble factors for bronchiolitis-associated deaths amid infants in the Us. Pediatr Infect Dis J. 2003, 22 (6): 483-490.
-
Boyce TG, Mellen BG, Mitchel EF, Wright PF, Griffin MR: Rates of hospitalization for respiratory syncytial virus infection amidst children in Medicaid. J Pediatr. 2000, 137 (6): 865-870. x.1067/mpd.2000.110531.
-
Nielsen HE, Siersma V, Andersen Southward, Gahrn-Hansen B, Mordhorst CH, Norgaard-Pedersen B, Roder B, Sorensen TL, Temme R, Vestergaard BF: Respiratory syncytial virus infection—risk factors for hospital admission: a case–control report. Acta Paediatr. 2003, 92 (11): 1314-1321. 10.1080/08035250310006304.
-
von Linstow ML, Hogh 1000, Nordbo SA, Eugen-Olsen J, Koch A, Hogh B: A customs study of clinical traits and run a risk factors for human metapneumovirus and respiratory syncytial virus infection during the outset year of life. Eur J Pediatr. 2008, 167 (10): 1125-1133. 10.1007/s00431-007-0643-4.
-
Nafstad P, Jaakkola JJ, Hagen JA, Botten G, Kongerud J: Breastfeeding, maternal smoking and lower respiratory tract infections. Eur Respir J. 1996, 9 (12): 2623-2629. 10.1183/09031936.96.09122623.
-
Carbonell-Estrany X, Quero J, Bustos G, Cotero A, Domenech E, Figueras-Aloy J, Fraga JM, Garcia LG, Garcia-Alix A, Del Rio MG, et al: Rehospitalization because of respiratory syncytial virus infection in premature infants younger than 33 weeks of gestation: a prospective study. IRIS Study Group. Pediatr Infect Dis J. 2000, 19 (7): 592-597. ten.1097/00006454-200007000-00002.
-
Carbonell-Estrany X, Quero J: Hospitalization rates for respiratory syncytial virus infection in premature infants born during two consecutive seasons. Pediatr Infect Dis J. 2001, 20 (9): 874-879. ten.1097/00006454-200109000-00010.
-
Law BJ, Langley JM, Allen U, Paes B, Lee DS, Mitchell I, Sampalis J, Walti H, Robinson J, O'Brien K, et al: The Pediatric Investigators Collaborative Network on Infections in Canada study of predictors of hospitalization for respiratory syncytial virus infection for infants built-in at 33 through 35 completed weeks of gestation. Pediatr Infect Dis J. 2004, 23 (9): 806-814. ten.1097/01.inf.0000137568.71589.bd.
-
Reeve CA, Whitehall JS, Buettner PG, Norton R, Reeve DM, Francis F: Predicting respiratory syncytial virus hospitalisation in Australian children. J Paediatr Kid Health. 2006, 42 (5): 248-252. 10.1111/j.1440-1754.2006.00849.x.
-
Bulkow LR, Singleton RJ, Karron RA, Harrison LH, Alaska RSVSG: Take chances factors for severe respiratory syncytial virus infection among Alaska native children. Pediatrics. 2002, 109 (2): 210-216. ten.1542/peds.109.2.210.
-
Office of Health and Ecology Assessment: Respiratory wellness furnishings of passive smoking: lung cancer and other disorders (EPA/600/6-90/006F). 1992, US Ecology Protection Agency, Washington, DC
-
Commission on Infectious Diseases: From the American Academy of Pediatrics: Policy statements–Modified recommendations for use of palivizumab for prevention of respiratory syncytial virus infections. Pediatrics. 2009, 124 (six): 1694-1701.
-
Moher D, Liberati A, Tetzlaff J, Altman DG: Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009, 6 (seven): e1000097-10.1371/journal.pmed.1000097.
-
The Cochrane Collaboration's tool for assessing chance of bias. http://www.ohg.cochrane.org/forms/Risk%20of%20bias%20assessment%20tool.pdf,
-
The Bear on-RSV Study Group: Palivizumab, a humanized respiratory syncytial virus monoclonal antibiotic, reduces hospitalization from respiratory syncytial virus infection in loftier-risk infants. Pediatrics. 1998, 102 (3 Pt i): 531-537.
-
Horn SD, Smout RJ: Result of prematurity on respiratory syncytial virus hospital resource use and outcomes. J Pediatr. 2003, 143 (five suppl): S133-S141.
-
Broughton S, Roberts A, Fox M, Pollina E, Zuckerman M, Chaudhry S, Greenough A: Prospective study of healthcare utilisation and respiratory morbidity due to RSV infection in prematurely born infants. Thorax. 2005, 60 (12): 1039-1044. 10.1136/thx.2004.037853.
-
Figueras-Aloy J, Carbonell-Estrany X, Quero-Jimenez J, Fernandez-Colomer B, Guzman-Cabanas J, Echaniz-Urcelay I, Domenech-Martinez E: FLIP-2 Study: risk factors linked to respiratory syncytial virus infection requiring hospitalization in premature infants born in Spain at a gestational historic period of 32 to 35 weeks. Pediatr Infect Dis J. 2008, 27 (9): 788-793. x.1097/INF.0b013e3181710990.
-
Figueras-Aloy J, Carbonell-Estrany X, Quero J: Case–control study of the gamble factors linked to respiratory syncytial virus infection requiring hospitalization in premature infants born at a gestational historic period of 33–35 weeks in Spain. Pediatr Infect Dis J. 2004, 23 (9): 815-820. ten.1097/01.inf.0000136869.21397.6b.
-
Holberg CJ, Wright AL, Martinez FD, Ray CG, Taussig LM, Lebowitz Doctor: Gamble factors for respiratory syncytial virus-associated lower respiratory illnesses in the showtime year of life. Am J Epidemiol. 1991, 133 (xi): 1135-1151.
-
Gurkan F, Kiral A, Dagli Due east, Karakoc F: The effect of passive smoking on the development of respiratory syncytial virus bronchiolitis. Eur J Epidemiol. 2000, sixteen (5): 465-468. 10.1023/A:1007658411953.
-
Hall CB, Hall WJ, Gala CL, MaGill FB, Leddy JP: Long-term prospective study in children after respiratory syncytial virus infection. J Pediatr. 1984, 105 (iii): 358-364. 10.1016/S0022-3476(84)80005-0.
-
Hayes EB, Hurwitz ES, Schonberger LB, Anderson LJ: Respiratory syncytial virus outbreak on American Samoa. Evaluation of risk factors. Am J Dis Kid. 1989, 143 (3): 316-321.
-
Stensballe LG, Kristensen Thousand, Simoes EA, Jensen H, Nielsen J, Benn CS, Aaby P: Atopic disposition, wheezing, and subsequent respiratory syncytial virus hospitalization in Danish children younger than 18 months: a nested case–control study. Pediatrics. 2006, 118 (five): e1360-e1368. 10.1542/peds.2006-0907.
-
Gavin NI, Leader S: Predictive accurateness of risk factors for RSV-related hospitalizations among infants in low-income families born at 32 to 35 weeks of gestation. J Clin Outcomes Manag. 2007, fourteen (half dozen): 323-331.
-
Carroll KN, Gebretsadik T, Griffin MR, Dupont WD, Mitchel EF, Wu P, Enriquez R, Hartert TV: Maternal asthma and maternal smoking are associated with increased risk of bronchiolitis during infancy. Pediatrics. 2007, 119 (6): 1104-1112. ten.1542/peds.2006-2837.
-
Koehoorn M, Karr CJ, Demers PA, Lencar C, Tamburic Fifty, Brauer Thousand: Descriptive epidemiological features of bronchiolitis in a population-based cohort. Pediatrics. 2008, 122 (6): 1196-1203. 10.1542/peds.2007-2231.
-
Marbury MC, Maldonado G, Waller L: The indoor air and children'south health study: methods and incidence rates. Epidemiology. 1996, vii (2): 166-174. 10.1097/00001648-199603000-00011.
-
McConnochie KM, Roghmann KJ: Parental smoking, presence of older siblings, and family unit history of asthma increase risk of bronchiolitis. Am J Dis Child. 1986, 140 (8): 806-812.
-
Groothuis JR, Gutierrez KM, Lauer BA: Respiratory syncytial virus infection in children with bronchopulmonary dysplasia. Pediatrics. 1988, 82 (2): 199-203.
-
Al-Shehri MA, Sadeq A, Quli One thousand: Bronchiolitis in Abha, Southwest Saudi Arabia: viral etiology and predictors for hospital admission. W Afr J Med. 2005, 24 (4): 299-304.
-
Somech R, Tal G, Gilad E, Mandelberg A, Tal A, Dalal I: Epidemiologic, socioeconomic, and clinical factors associated with severity of respiratory syncytial virus infection in previously healthy infants. Clin Pediatr (Phila). 2006, 45 (seven): 621-627. 10.1177/0009922806291012.
-
Al-Sonboli N, Hart CA, Al-Aghbari N, Al-Ansi A, Ashoor O, Cuevas LE: Human metapneumovirus and respiratory syncytial virus disease in children, Republic of yemen. Emerg Infect Dis. 2006, 12 (9): 1437-1439.
-
Bradley JP, Bacharier LB, Bonfiglio J, Schechtman KB, Strunk R, Storch 1000, Castro G: Severity of respiratory syncytial virus bronchiolitis is affected by cigarette smoke exposure and atopy. Pediatrics. 2005, 115 (i): e7-e14.
-
Chatzimichael A, Tsalkidis A, Cassimos D, Gardikis S, Tripsianis Yard, Deftereos S, Ktenidou-Kartali S, Tsanakas I: The role of breastfeeding and passive smoking on the development of astringent bronchiolitis in infants. Minerva Pediatr. 2007, 59 (3): 199-206.
-
Sritippayawan S, Prapphal Northward, Wong P, Tosukhowong P, Samransamruajkit R, Deerojanawong J: Environmental tobacco smoke exposure and respiratory syncytial virus infection in young children hospitalized with acute lower respiratory tract infection. J Med Assoc Thai. 2006, 89 (12): 2097-2103.
-
Reese Air conditioning, James IR, Landau LI, Lesouef PN: Relationship between urinary cotinine level and diagnosis in children admitted to hospital. Am Rev Respir Dis. 1992, 146 (1): 66-70.
-
Benowitz NL, Jacob P: Nicotine and cotinine emptying pharmacokinetics in smokers and nonsmokers. Clin Pharmacol Ther. 1993, 53 (iii): 316-323. 10.1038/clpt.1993.27.
-
Tobacco use among U.Due south. racial/ethnic minority groups—African Americans, American Indians and Alaska Natives, Asian Americans and Pacific Islanders, Hispanics. A Study of the Surgeon General. Executive summary. MMWR Recomm Rep. 1998, 47 (RR-xviii): v-15. 1–16
-
Windham GC, Swan SH, Fenster L: Parental cigarette smoking and the risk of spontaneous abortion. Am J Epidemiol. 1992, 135 (12): 1394-1403.
-
Office of the Surgeon General: The health consequences of smoking: a written report of the Surgeon General. Edited by Services UDoHaH. Washington, DC. 2004, U.s.a. Section of Health and Human Services
Pre-publication history
-
The pre-publication history for this newspaper can be accessed here:http://www.biomedcentral.com/1471-2431/12/81/prepub
Acknowledgements
Editorial assistance was provided by Susan DeRocco, PhD, and Gerard P. Johnson, PhD, of Complete Healthcare Communications, Inc. (Chadds Ford, PA) and was funded by MedImmune.
Writer information
Affiliations
Corresponding author
Additional information
Competing interests
This projection was funded under a contract with MedImmune, LLC. Joseph R. DiFranza, Physician, provided consultant services to MedImmune. Amy Thou. Barrett, MSPH, MA, and Ann D. Colosia, PhD, are employees of RTI Health Solutions and provided consulting services to MedImmune. Parthiv J. Mahadevia, Doctor, MPH, and Anthony Masaquel, PhD, MPH, are employees of MedImmune. All authors agreed on the concluding text and conclusions of the manuscript. In that location are no other competing interest disclosures.
Authors' contributions
JD participated in the conception of the literature review and paper, reviewed published studies, analyzed data, and drafted portions of the manuscript. AM participated in the conception of the literature review and paper, reviewed studies, analyzed information, and critically revised the manuscript for of import intellectual content. AB and AC each participated in the design and conception of the literature review and paper, reviewed published studies, extracted and analyzed data, and drafted portions of the manuscript. PM participated in the conception of the manuscript, analyzed data, and critically revised the manuscript for important intellectual content. All authors read and canonical the terminal manuscript.
Electronic supplementary cloth
Authors' original submitted files for images
Rights and permissions
This article is published under license to BioMed Central Ltd. This is an Open Admission article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted utilise, distribution, and reproduction in whatever medium, provided the original work is properly cited.
Reprints and Permissions
Almost this article
Cite this article
DiFranza, J.R., Masaquel, A., Barrett, A.M. et al. Systematic literature review assessing tobacco smoke exposure as a risk cistron for serious respiratory syncytial virus disease among infants and immature children. BMC Pediatr 12, 81 (2012). https://doi.org/x.1186/1471-2431-12-81
-
Received:
-
Accepted:
-
Published:
-
DOI : https://doi.org/10.1186/1471-2431-12-81
Keywords
- Respiratory syncytial virus
- Respiratory infection
- Tobacco
- Smoking
- Children
hendersonnevency65.blogspot.com
Source: https://bmcpediatr.biomedcentral.com/articles/10.1186/1471-2431-12-81
Post a Comment for "Baby Has Rsv Could Mother Using Vapor Cigarette"